
Concept explainers
(a)
Interpretation:
The second resonance structure of the following ion should be drawn:
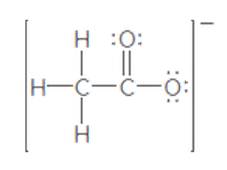
Concept Introduction:
When more than one Lewis structure can be drawn for a molecule or ion, then it is said to have resonance.
It is generally the delocalization of electrons (bonds) over three or more atoms in a molecule for which one simple Lewis structure cannot depicts its correct structure.
(b)
Interpretation:
The second resonance structure of the following ion should be drawn:
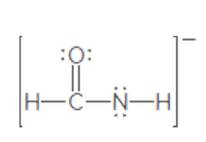
Concept Introduction:
When more than one Lewis structure can be drawn for a molecule or ion, then it is said to have resonance.
It is generally the delocalization of electrons (bonds) over three or more atoms in a molecule for which one simple Lewis structure cannot depicts its correct structure.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Draw two resonance structures of the given molecule/ion. Be sure to include all lone pairs. :Ö:arrow_forwardDraw a second resonance structure for each species in parts (a), (b), and (c). Draw two additional resonance structures for the ion in part (d).arrow_forwardDraw an equivalent resonance structure that minimizes charge. Include all lone pairs in your structure.arrow_forward
- Draw one valid Lewis structure for each compound. Assume the atomsare arranged as drawn.arrow_forwardDraw an equivalent resonance structure that minimizes charge. Include all lone pairs in your structure. OU Harrow_forwardDraw two resonance structures of the given molecule/ion. Be sure to include all lone pairs. + NHarrow_forward
- Draw an equivalent resonance structure that minimizes charges. Include all lone pairs in your structure.arrow_forwardNitrate ion, how many non bonding lone pairs are in ion ? Molecular shape and how many resonance structure can be drawn from the ion ?arrow_forwardDraw the resonance structure indicated by the curved arrows. Assign formal charges. H H/:O: H H-C-C-C-Ċ—H H H H Draw the molecule by placing atoms on the canvas and connecting them with bonds. Include all hydrogen atoms and nonbonding electrons. Show the formal charges of all atoms in the correct structure.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





