
Concept explainers
(a)
Interpretation:
The structure formula with all valence electrons should be drawn for a carbonyl group.
Concept Introduction:
Lewis structures can be drawn by following some point for any compound as mentioned below:
- Find out number of valence electrons present in the molecule
- The connectivity of atoms need to be understood like which atoms are linked with each other and connect them with covalnet bonds
- The arrangements of electrons in proper manner to achieve complete outer shell
Answer to Problem 23P
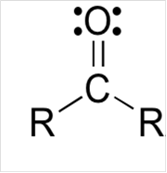
Lewis structure for a carbonyl group:
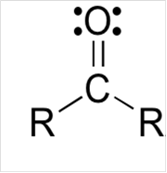
Explanation of Solution
The structure of any atom of prepared with the help of Lewis model which explains guideline for bonding of atom. It guides about the covalent bonds which are formed in various combinations of single, double and triple bonds.
Single covalent bond occurs by sharing one pair of electrons and can be represented by single line in between atoms.
Double and triple bonds are formed when two and three pairs of electrons are shared in between atoms respectively and can be represented by two or three lines in between the atoms.
For several covalent bonds in organic compound containing carbon bonded to hydrogen, nitrogen, chlorine and oxygen shows some important points to be noted for structure are as follow:
- Carbon forms four covalent bonds while hydrogen form one covalent bond and both has no unshared pair of electrons.
- Three covalent bonds are formed in nitrogen atom with only one unshared pair of electron left behind.
- Oxygen can form two covalent bonds and have further more two unshared pair of electron.
- Iodine, chlorine, bromine and fluorine like halogens can form only one covalent bond like hydrogen but they have three unshared pair of electrons.
To draw Lewis structure for a carbonyl group, following are the steps:
(b)
Interpretation:
The structure formula with all valence electrons should be drawn for a carboxyl group.
Concept Introduction:
Lewis structures can be prepared by following some point for any compound as mentioned below:
- Find out number of valence electrons present in the molecule
- The connectivity o atoms need to be understood like which atoms are linked with each other and connect them with covalnet bonds
- The arrangements of electrons in proper manner to achieve complete outer shell
Answer to Problem 23P
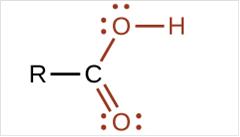
To draw Lewis structure for a carboxyl group:
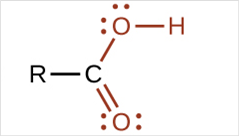
Explanation of Solution
The structure of any atom of prepared with the help of Lewis model which explains guideline for bonding of atom. It guides about the covalent bonds which are formed in various combinations of single, double and triple bonds.
Single covalent bond occurs by sharing one pair of electrons and can be represented by single line in between atoms.
Double and triple bonds are formed when two and three pairs of electrons are shared in between atoms respectively and can be represented by two or three lines in between the atoms.
For several covalent bonds in organic compound containing carbon bonded to hydrogen, nitrogen, chlorine and oxygen shows some important points to be noted for structure are as follow:
- Carbon forms four covalent bonds while hydrogen form one covalent bond and both has no unshared pair of electrons.
- Three covalent bonds are formed in nitrogen atom with only one unshared pair of electron left behind.
- Oxygen can form two covalent bonds and have further more two unshared pair of electron.
- Iodine, chlorine, bromine and fluorine like halogens can form only one covalent bond like hydrogen but they have three unshared pair of electrons.
To draw Lewis structure for a carboxyl group, following are the steps:
(c)
Interpretation:
The structure formula with all valence electrons should be drawn for a hydroxyl group.
Concept Introduction:
Lewis structures can be prepared by following some point for any compound as mentioned below:
- Find out number of valence electrons present in the molecule
- The connectivity o atoms need to be understood like which atoms are linked with each other and connect them with covalnet bonds
- The arrangements of electrons in proper manner to achieve complete outer shell
Answer to Problem 23P
To draw Lewis structure for a hydroxyl group:
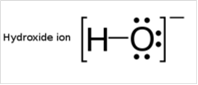
Explanation of Solution
The structure of any atom of prepared with the help of Lewis model which explains guideline for bonding of atom. It guides about the covalent bonds which are formed in various combinations of single, double and triple bonds.
Single covalent bond occurs by sharing one pair of electrons and can be represented by single line in between atoms.
Double and triple bonds are formed when two and three pairs of electrons are shared in between atoms respectively and can be represented by two or three lines in between the atoms.
For several covalent bonds in organic compound containing carbon bonded to hydrogen, nitrogen, chlorine and oxygen shows some important points to be noted for structure are as follow:
- Carbon forms four covalent bonds while hydrogen form one covalent bond and both has no unshared pair of electrons.
- Three covalent bonds are formed in nitrogen atom with only one unshared pair of electron left behind.
- Oxygen can form two covalent bonds and have further more two unshared pair of electron.
- Iodine, chlorine, bromine and fluorine like halogens can form only one covalent bond like hydrogen but they have three unshared pair of electrons.
To draw Lewis structure for a hydroxyl group, following are the steps:
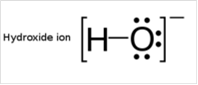
(d)
Interpretation:
The structure formula with all valence electrons should be drawn for a primary
Concept Introduction:
Lewis structures can be prepared by following some point for any compound as mentioned below:
- Find out number of valence electrons present in the molecule
- The connectivity o atoms need to be understood like which atoms are linked with each other and connect them with covalnet bonds
- The arrangements of electrons in proper manner to achieve complete outer shell
Answer to Problem 23P
To draw Lewis structure for a primary amino group:
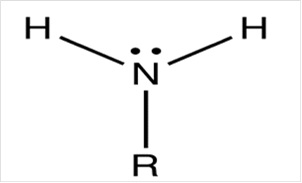
Explanation of Solution
The structure of any atom of prepared with the help of Lewis model which explains guideline for bonding of atom. It guides about the covalent bonds which are formed in various combinations of single, double and triple bonds.
Single covalent bond occurs by sharing one pair of electrons and can be represented by single line in between atoms.
Double and triple bonds are formed when two and three pairs of electrons are shared in between atoms respectively and can be represented by two or three lines in between the atoms.
For several covalent bonds in organic compound containing carbon bonded to hydrogen, nitrogen, chlorine and oxygen shows some important points to be noted for structure are as follow:
- Carbon forms four covalent bonds while hydrogen form one covalent bond and both has no unshared pair of electrons.
- Three covalent bonds are formed in nitrogen atom with only one unshared pair of electron left behind.
- Oxygen can form two covalent bonds and have further more two unshared pair of electron.
- Iodine, chlorine, bromine and fluorine like halogens can form only one covalent bond like hydrogen but they have three unshared pair of electrons.
To draw Lewis structure for a carbonyl group, following are the steps:
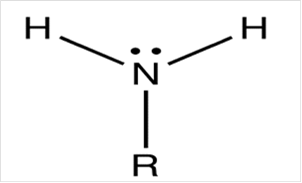
(e)
Interpretation:
The structure formula with all valence electrons should be drawn for an ester group.
Concept Introduction:
Lewis structures can be prepared by following some point for any compound as mentioned below:
- Find out number of valence electrons present in the molecule
- The connectivity o atoms need to be understood like which atoms are linked with each other and connect them with covalnet bonds
- The arrangements of electrons in proper manner to achieve complete outer shell
Answer to Problem 23P
To draw Lewis structure for an ester group:
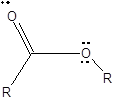
Explanation of Solution
The structure of any atom of prepared with the help of Lewis model which explains guideline for bonding of atom. It guides about the covalent bonds which are formed in various combinations of single, double and triple bonds.
Single covalent bond occurs by sharing one pair of electrons and can be represented by single line in between atoms.
Double and triple bonds are formed when two and three pairs of electrons are shared in between atoms respectively and can be represented by two or three lines in between the atoms.
For several covalent bonds in organic compound containing carbon bonded to hydrogen, nitrogen, chlorine and oxygen shows some important points to be noted for structure are as follow:
- Carbon forms four covalent bonds while hydrogen form one covalent bond and both has no unshared pair of electrons.
- Three covalent bonds are formed in nitrogen atom with only one unshared pair of electron left behind.
- Oxygen can form two covalent bonds and have further more two unshared pair of electron.
- Iodine, chlorine, bromine and fluorine like halogens can form only one covalent bond like hydrogen but they have three unshared pair of electrons.
To draw Lewis structure for an ester group, following are the steps:
Want to see more full solutions like this?
Chapter 10 Solutions
Introduction To General, Organic, And Biochemistry
- With reference to organic molecules, explain the following structure: CH2 CH— О CH2arrow_forwardWhat does a functional group do in an organic molecule?arrow_forwardHow many electron pairs are shared when a triple bond exists between two carbon atoms? What must he the geometric arrangement around the carbon atoms in a triple bond? Draw the Lewis structure of a simple molecule that contains a triple bond.arrow_forward
- The functional group is the center of reactivity in an organic molecule. Identify the functional groups in the ff. structures. If a functional group is found more than once, it need be to be circled once.arrow_forwardComplete the Lewis structure for the following condensed structural formula. CH3COCOOHarrow_forwardWrite the systematic name of each organic molecule: structure s name 1 0 0 X 5arrow_forward
- We see that 1-propanol and 2-propanol have the same molecular formula, C3H7OH, but different molecular structures. What is the name for molecules that have the same molecular formula but different structural formulas (different shapes)? Use the specific term.arrow_forwardThe clear formula of an active drug substance is given below. Write the closed formula of this compound, it consists of the combination of two organic compounds that we have seen so far. Write the name of these two structures.arrow_forwardShown below is the skeletal structure of a commonly used artificial sweetener. Which functional groups are found in this structure? Select all that apply. CI- HO CI Ether O Aldehyde O Ketone O Amide Alcohol Amine O OH Ester Carboxylic Acid O OH OH OHarrow_forward
- Write the systematic name of each organic molecule: HO HO structure name OH ☐ OH Uarrow_forwardWhich statement describes a functional group? O an inert group of atoms that is part of a larger molecule molecule that can participate in a chemical reaction O any the backbone of a macromolecule, such as a carbohydrate O part of a larger molecule that can react with other atoms or moleculesarrow_forwardGive the definition of a covalent bond. State WHY covalent bonds form in organic molecules.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning




